Author Guidelines
INAVA welcomes contributions that promote the exchange of ideas and rational discourse between practicing educators and medical researchers worldwide. Specific topics of interest include all aspects of medical and physiology, modeling of diagnosing and cure of human diseases, the scientific underpinning of medicine and pathophysiology, biochemistry and biology molecular solution, decision support systems, biomedicine, and any experimental design and statistical analysis, the application of new analytical and study methods (including biomolecular studies). Articles should be written in clear, concise English following scientific writing recommendations found in Scientific Style and Format, the Council of Science Editors (CSE) style manual (7th ed., 2006, Reston, VA, Council of Science Editors). Only material that has not been published previously (either in print or electronically) and is not under consideration for publication elsewhere, except for an abstract published in conference proceedings, will be considered for publication. Prior presentation of data (i.e., at a scientific meeting) does not preclude publication in INAVA. All submissions to the journal will be scanned for possible duplicate or prior publication using the Turnitin plagiarism detection system. Authors who submit previously published work to the journal will be banned from submitting future manuscripts to the journal, and their funding body and/or institution will be notified.
To summarize everything, here is a template that you may download as a solid guide for editing your manuscripts:
Template INAVA (Original Article).
Template INAVA (Letter to Editor).
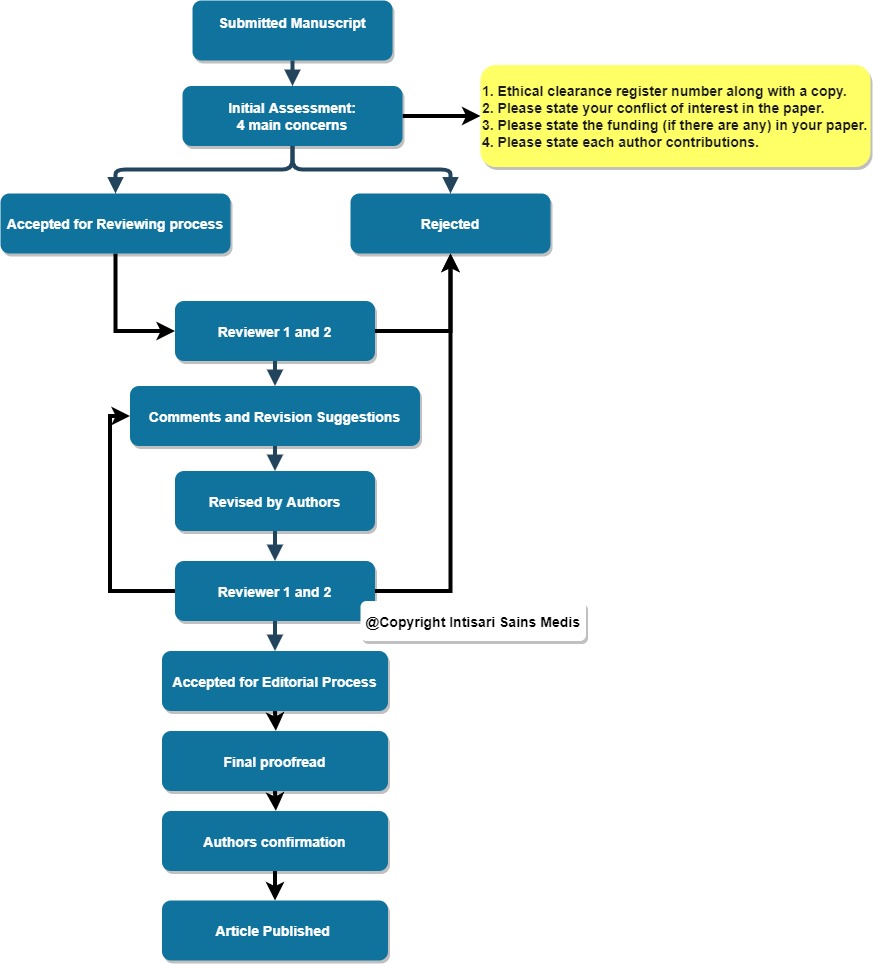
Before start editing based on our Author and Review Guidelines, these are the four-items that we want our author to prepare and clearly state in the manuscripts:
- Ethical clearance register number along with a copy. If your work is a case series or case report, please send us the patient/family informed consent (in English), and fill out the ICJME form that can be found below.
2. Please state your conflict of interest in the paper.
3. Please state the funding (if there are any) in your paper. (Form 2 and 3 Download ICJME form here)
4. Please state each author's contributions. (Download here)
(Without these four items, your manuscript will automatically be rejected).
Double-Blind Peer Review Guidelines
INAVA uses double-blind review, which means that both the reviewer and author identities are concealed from the reviewers, and vice versa, throughout the review process.
- Submission
To facilitate the double-blind review, authors need to ensure that their manuscripts are prepared in a way that does not give away their identity. To help with this preparation, please ensure the following when submitting to INAVA:
- Submit the Title Page containing the Authors details and Blinded Manuscript with no author details as 2 separate files.
Information to help prepare the Title Page
This should include the title, authors' names and affiliations, and a complete address for the corresponding author, including telephone and e-mail address.
Information to help prepare the Blinded Manuscript
Besides the obvious need to remove names and affiliations under the manuscript's title, other steps need to be taken to ensure the manuscript is correctly prepared for double-blind peer review. To assist with this process, the key items that need to be observed are as follows:
- Use the third person to refer to work the Authors have previously undertaken, e.g., replace any phrases like “as we have shown before” with “… has been shown before [Anonymous, 2007]”.
- Make sure figures do not contain any affiliation related identifier.
- Do not eliminate essential self-references or other references but limit self-references only to papers relevant to those reviewing the submitted paper.
- Cite papers published by the Author in the text as follows: ‘[Anonymous, 2007]’.
- For blinding in the reference list: ‘[Anonymous 2007] Details omitted for double-blind reviewing.’
- Remove references to funding sources.
- Do not include acknowledgments.
- Remove any identifying information, including author names, from file names and ensure document properties are also anonymized.
- Initial Assessment
The manuscript will undergo an initial assessment to check the manuscript topic related to the journal aims and scope, grammar quality, and the 4 requirement items described above.
- Peer Review
After passing the initial check, the manuscript will be reviewed by 2 reviewers acknowledged on their field of expertise and certified by Publons (Web of Science) to ensure the quality of the comments and suggestions.
In addition to our reviewer guidelines, we also encourage our reviewer to use a checklist when accessing a manuscript, either using Publons checklist or other specific checklists related to the article type.
We have collected and regularly update the reviewer checklist on the following link:
The reviewer may decide either to reject the article or to comments on some suggestion and clarify any parts of the article assigned to them.
The authors will have 7 days to reply and answer the reviewers' questions and inquiry and resend it to the system. Once again, the reviewer will check the manuscript and give their final decisions (Accept/ Reject/ Revisions Required).
- Editorial Process
And if the article passed the reviewing process, it will be handled to the editor for final proofread and editing process. The author needs to confirm the final version for publications and add/revise minor parts of the article based on the editor's suggestions before the article is converted to the pdf galley and published on the website.
Authorship
Authorship should be based on:
- Substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data
- Drafting the article or revising it critically for important intellectual content
- Final approval of the version to be published
- Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
All persons designated as authors should qualify for authorship, and all those who qualify should be listed. Each author should have participated sufficiently to take public responsibility for appropriate portions of the content. Acquisition of funding, data collection, or general supervision of the research group alone does not constitute authorship.
When a large, multicenter group has conducted the work, the group should identify the individuals who accept direct responsibility for the manuscript. These individuals should fully meet the authorship criteria defined above, and editors will ask these individuals to complete journal-specific author and conflict-of-interest disclosure forms.
All contributors who do not meet the criteria for authorship should be listed in an Acknowledgments section. Examples of those who might be acknowledged include a person who provided purely technical help, writing assistance, or a department chairperson who provided only general support.
The journal currently does not permit the changing/adding/deleting of authors after submitting the paper. Changing the author sequence after the manuscript is submitted is also prohibited. So write down the author sequence wisely. Those who made the greatest contribution are generally listed first, and if all co-authors contribute similarly in the manuscript, alphabetical order may also be used.
Manuscript Format
The manuscript should accompany a title page, which includes a short running title; the first name, middle initial, and last name of each author; the affiliation (in English) of each author during the study being reported; the name, current address, telephone number, fax number, and e-mail address of the corresponding author; and the word count and the number of tables and figures. The text on the title page should be center aligned. The main text and tables must be saved in Microsoft Word document format, with 12 pt Times New Roman font, and the main text should double-spaced with justified margins. Please do not use headers, footers, or endnotes in your paper.
Research Articles
Research Articles are expected to present a significant advance in the areas of pre-clinical medicine or/and clinical medicine and arranged in the following order: title page, abstract, introduction (no heading necessary), "Research Design and Methods," Results," "Discussion," "Acknowledgments," "References," tables (each including a title and legend), figure legends, and figures.
- An abstract is required for all articles. The abstract should be unstructured, concise, and precise and should convey the manuscript's essential findings. The abstract may not exceed 300 words. It should state the rationale, objectives, findings, and conclusions of the manuscript. References, primary data, and statistical significance should not be presented in the abstract, and nonstandard abbreviations must be defined.
- The article should contain no more than 25 references, and the reference section should be single-spaced with justified margins. The references should also include the DOI number from CrossRef.
Review Articles
Review articles are comprehensive, critical reviews of research topics and/or methodological approaches that are highly relevant to investigators in medical research. The review should be interpretative rather than a summary that describes various available approaches and their relative merits, limitations, and specific applications.
Tables
Tables should be double-spaced on separate pages and included at the end of the text document, with the table number and title indicated. Tables should be created using Word and the "Insert Table" command; please do not use tabs and/or spaces to create tables, columns, or rows. Tables with internal divisions (Tables 1A and B) should be submitted as individual tables, i.e., Tables 1 and 2. Symbols for units should be confined to column headings. Abbreviations should be kept to a minimum and defined in the table legend. Please avoid the use of shading. If a table includes data that requires an explanation in the legend, apply the following symbol sequence, from top to bottom, left to right: *, †, ‡, §, ||, ¶, #, **, ††, ‡‡, etc.
If tables are taken from other sources, it should be noted in the legend. The author must be able to provide written permission for reproduction obtained from the original publisher and author.
Figures
INAVA uses digital publishing methods throughout the journal production process. If your article is accepted, it will be published online. The following sections provide information on how to format your figures to ensure your images' best possible reproduction.
Size - Figures should be produced at the size they are to appear in the printed journal. Please make sure your figures will fit in one or two columns in width. Multi-paneled figures should be assembled in a layout that leaves the least amount of blank space.
Font - At 100% size, fonts should be 8-10 points and used consistently throughout all figures.
Text - Information on the axes should be succinct, using abbreviations where possible, and the label on the y-axis should read vertically, not horizontally. Key information should be placed in any available white space within the figure; if space is not available, it should be placed in the legend. In general, figures with multiple parts should be marked A, B, C, etc., with a description of each panel included in the legend rather than on the figure.
Line and bar graphs - Lines in graphs should be bold enough to be easily read after reduction, as should all symbols used in the figure. Data points are best marked with the following symbols, again assuring that they will be readily distinguishable after reduction. In the figure legend, please use words rather than the symbols; e.g., "black circles = group 1; white squares = group 2; black bars = Hb1AC; white bars = glucose levels." Bars should be black or white only unless more than two datasets are presented; additional bars should be drawn with clear, bold hatch marks or stripes, not shades of grey.
Line or bar graphs or flow charts with text should be created in black and white, not shades of grey, difficult to reproduce in even tones.
Reproductions - If materials (e.g., figures and/or tables) are taken from other sources, it should be noted in the legend. The author must be able to provide written permission for reproduction obtained from the original publisher author.
Figure legends. Figure legends should be clearly numbered and included at the end of your document and should not be included in the separate figure/image files. Please use words to describe symbols used in the figure; e.g., "black circles = group 1; white squares = group 2; black bars = Hb1AC; white bars = glucose levels."
Abbreviations
Abbreviations should be used only when necessary, e.g., long chemical names (HEPES), procedures (ELISA), or terms used throughout the article. Abbreviate units of measure only when used with numbers. Abbreviations may be used in tables and figures.
Units
Clinical laboratory values should be in the International System of Units (SI) form. Kilocalories should be used rather than kilojoules. HbA1c values should be dually reported as “% (mmol/mol).” Please use the NGSP’s HbA1c converter at http://www.ngsp.org/convert1.asp to calculate HbA1c values as both % and mmol/mol.
References
References should be listed according to the Vancouver Style (Thomas MC. Diuretics, ACE inhibitors, and NSAIDs – the triple whammy. Med J Aust. 2000;172:184–185 http://dx.doi.org/10.1006/jmbi.1995.0238; Guilbert TW, Morgan WJ, Zeiger RS, Mauger DT, Boehmer SJ, Szefler SJ, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med. 2006;354:1985–97 http://dx.doi.org/........; http://www.icmje.org/ )and should be numbered in the order that they are cited in the text. All authors must be listed and inclusive page numbers provided. Journal titles should be abbreviated as in the National Library of Medicine's List of Journals Indexed for Medline; for unlisted journals, complete journal titles should be provided.
Failure to follow instructions may result in publication delays if your manuscript is accepted.
